Pioneering Regulatory Excellence in Saudi Arabia
Welcome to UniFusion B.S.C, a premier regulatory consultation firm specializing in Medical Devices, In Vitro Diagnostics (IVDs), Cosmetics, and Drug registration in Saudi Arabia. With deep expertise and an unwavering commitment to compliance, we navigate the complexities of regulatory frameworks to streamline approvals and ensure seamless market access.
As trusted advisors, we work closely with the Saudi Food and Drug Authority (SFDA), offering unparalleled guidance tailored to your business needs. Whether you’re launching innovative healthcare solutions or expanding product portfolios, our team delivers precision-driven strategies that meet Saudi regulatory standards with efficiency and integrity.
At UniFusion B.S.C, we don’t just offer consultation—we empower progress, ensuring your products meet the highest safety and quality benchmarks while accelerating your path to success in Saudi Arabia’s dynamic healthcare landscape.
Our services
Our clients
Our news
UniFusion Featured in Riyadh Daily
UniFusion B.S.C. is proud to be featured in Riyadh Daily, highlighting its growth and strategic outlook within Saudi Arabia’s evolving investment landscape. The article reflects the Kingdom’s position as a stable and resilient hub for long-term investment, even amid global uncertainty.
Over the past two years, UniFusion has achieved strong growth by supporting international medical device companies entering the Saudi market. As the company moves forward, it is focused on strengthening its operations, enhancing its corporate culture, and building long-term partnerships.
UniFusion reaffirms its commitment to delivering reliable regulatory support and contributing to the advancement of healthcare and regulatory excellence in line with Vision 2030.
VertiGoggles® Receives SFDA Medical Device Marketing Authorization
19.05.2026
We are proud to announce the successful Medical Device Marketing Authorization (MDMA) approval from the Saudi Food and Drug Authority (SFDA) for the innovative VertiGoggles® system by Shanghai ZEHNIT Medical Technology Co., Ltd.
VertiGoggles® is an advanced binocular infrared diagnostic system designed to support comprehensive vestibular and balance assessment by combining Video Nystagmography (VNG) and true Video Head Impulse Testing (vHIT) within a single integrated platform. The system offers precise 3D tracking, reliable diagnostic data, and a lightweight plug-and-play design that enhances clinical efficiency and patient care.
Through this achievement, UniFusion B.S.C. continues its commitment to supporting global medical device manufacturers in obtaining SFDA regulatory approvals and expanding access to innovative healthcare technologies within the Saudi market.
UniFusion at WHX Dubai 2026 (Formerly Arab Health)
The UniFusion team was pleased to visit WHX Dubai 2026, formerly known as Arab Health, taking place from 9–12 February 2026 at the Dubai Exhibition Centre, Expo City Dubai.
WHX Dubai marks the evolution of one of the world’s most influential healthcare exhibitions, bringing together global leaders, innovators, manufacturers, and regulators to shape the future of healthcare across medical devices, pharmaceuticals, diagnostics, and digital health.
During the event, the UniFusion team actively engaged with industry stakeholders, providing expert support as Regulatory Affairs Consultants. Our discussions focused on regulatory strategy, compliance pathways, and market access for medical devices and pharmaceutical products, with particular attention to GCC and international regulatory requirements.
Our presence at WHX Dubai reflects UniFusion’s continued commitment to helping healthcare companies navigate complex regulatory landscapes and accelerate compliant market entry with confidence.
UniFusion BSC at WHX Labs Dubai 2026
The UniFusion BSC team is proud to be attending WHX Labs Dubai 2026 (formerly known as Medlab Middle East), taking place at the Dubai World Trade Centre, UAE, from 10–13 February 2026.
WHX Labs remains one of the most influential global platforms for laboratory medicine, diagnostics, and healthcare innovation. The event brings together manufacturers, regulatory experts, laboratory professionals, and healthcare leaders to explore advancements that are shaping the future of patient care worldwide.
On Day 2, our team continues to engage with global manufacturers and industry partners, discussing the latest technologies and regulatory developments that are transforming the diagnostics landscape.
The 3rd International Conference on Clinical Trials (ICCT 2026)
UniFusion attended The 3rd International Conference on Clinical Trials (ICCT 2026), held on 4–5 February 2026 at Narcissus The Royal Hotel, Riyadh, Saudi Arabia.
ICCT 2026 provided an international forum for researchers, clinicians, industry leaders, and healthcare professionals to engage in meaningful dialogue on emerging trends, innovations, and challenges in clinical trials. Organized around the theme “Voice of Hope,” the conference featured high-level scientific sessions, expert-led discussions, and strategic networking opportunities that underscored ongoing advancements in clinical research.
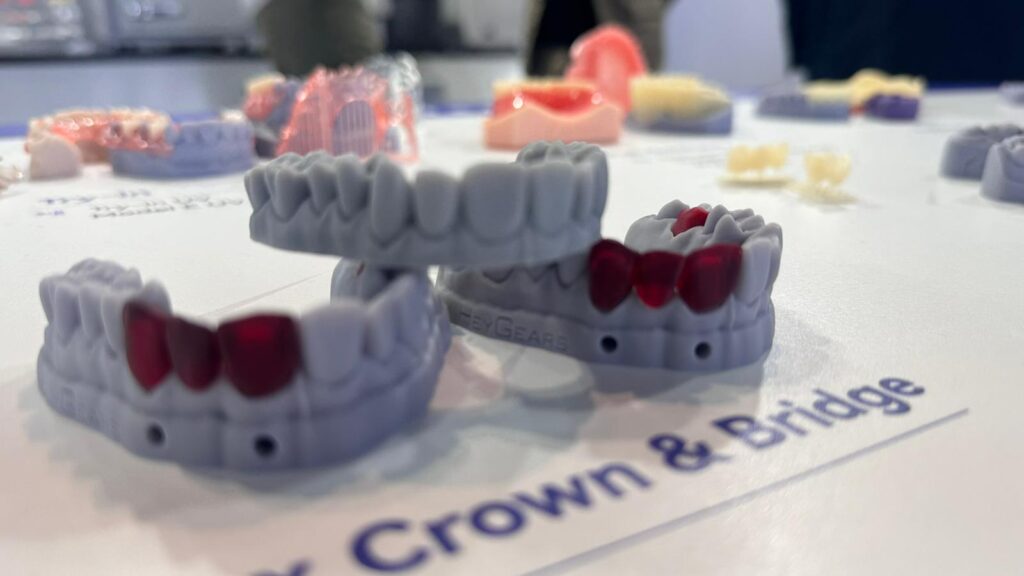
The 37th Saudi International Dental Conference – Riyadh 2026
UniFusion B.S.C was proud to be present in Riyadh during the 37th Saudi International Dental Conference (SIDC Riyadh 2026), one of the region’s leading platforms for dental innovation and medical device advancement.
Held from February 5–7, 2026, at the Riyadh Front Exhibition & Conference Center, SIDC brought together international manufacturers, regulators, industry leaders, and healthcare professionals to explore emerging technologies, regulatory developments, and the future direction of the dental and medical device sectors.
AEEDC Dubai 2026 — A Powerful Start for UniFusion B.S.C
UniFusion B.S.C was proud to participate in AEEDC Dubai 2026, held at the Dubai World Trade Centre, one of the world’s largest annual dental conferences and exhibitions.
The event brought together global industry leaders, manufacturers, and decision-makers, providing an excellent platform for UniFusion to engage with valued clients, connect with international partners, and explore the latest innovations shaping the dental and healthcare industry. Throughout the exhibition, our team focused on strengthening partnerships, discussing emerging technologies, and identifying new collaboration opportunities.
AEEDC Dubai 2026 reinforced UniFusion’s commitment to innovation, growth, and long-term collaboration across the region and beyond.
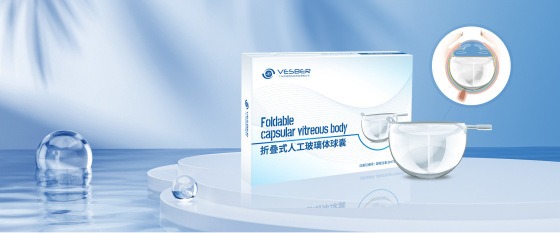
Unifusion BSC Achieves MDMA Approval for the World’s First Foldable Capsular Vitreous Body (FCVB)
Unifusion BSC has achieved a major regulatory milestone by obtaining SFDA Marketing Authorization for the Foldable Capsular Vitreous Body (FCVB), manufactured by Guangzhou Vesber Biotechnology Co., Ltd. The FCVB is the world’s first safe and effective artificial vitreous substitute designed to closely mimic the natural human vitreous body. This innovative technology provides 360-degree retinal support, prevents silicone oil-related complications, preserves intraocular function, and offers a critical new treatment option for patients with severe ocular trauma, silicone oil-dependent eyes, and complex retinal detachments. The approval reflects Unifusion BSC’s commitment to advancing healthcare innovation in the Kingdom in line with Saudi Vision 2030.
UniFusion B.S.C at MEDICA 2025 – Germany
UniFusion B.S.C visited the MEDICA Exhibition in Düsseldorf from 17 to 20 November 2025. During the event, our team engaged with global innovators, industry specialists, and leading medical device manufacturers to explore the latest healthcare technologies and emerging trends. The visit provided valuable opportunities to strengthen partnerships, discuss regulatory pathways, and identify advanced solutions that support efficient and compliant market entry. This engagement aligns with UniFusion’s commitment to creating a fusion of innovation, compliance, and market growth for medical device companies worldwide.
UniFusion B.S.C at Global Health Exhibition 2025 – Riyadh
UniFusion B.S.C proudly participated in the Global Health Exhibition, held in Riyadh from October 27–30, 2025. Our team had the honor of meeting H.E. Dr. Hisham bin Saad Aljadhey, CEO of the SFDA, along with other key leaders to discuss innovation and regulatory collaboration. Throughout the event, we connected with global partners, explored new opportunities, and reinforced our role as a trusted bridge between international medical device manufacturers and the Saudi FDA. UniFusion continues to lead in advancing regulatory excellence and empowering global healthcare compliance.
UniFusion B.S.C Partners with United Imaging Healthcare
We’re pleased to announce our partnership with United Imaging Healthcare (UIH), a global leader in advanced medical imaging. Operating in over 75 countries, UIH has delivered more than 30,000 high-performance systems, supporting healthcare providers with innovative solutions. Through this partnership, UniFusion B.S.C will facilitate UIH’s regulatory compliance and market access in Saudi Arabia, strengthening our mission to bring world-class medical technologies to the region.
UniFusion at the 91st CMEF in Shanghai
UniFusion proudly participated in the 91st China International Medical Equipment Fair (CMEF), held from April 8–11, 2025, in Shanghai.
Our Regulatory Portfolio Director, Mr. Mohiuddin Farooqui, delivered a live seminar on the SFDA Registration Process, guiding manufacturers on entering the Saudi market. CMEF 2025 is a valuable opportunity to showcase our expertise and connect with global healthcare innovators.
contact us
stay updated
Stay Ahead with Expert Insights—Subscribe for the Latest in Regulatory News